Water alkalinity and pH are not the same. Water pH measures the amount of hydrogen (acid ions) in the water, whereas water alkalinity is a measure of the carbonate and bicarbonate levels in water. Think of carbonates and bicarbonates as dissolved limestone. The higher the alkalinity of the water, the more lime it contains and therefore, the more rapidly the water can cause the growing medium pH to rise. On the other hand, the pH of the water does not have any influence on the pH of the growing medium.
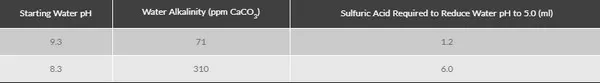
For example, the chart above shows the starting pH of two water sources and the amount of acid required for each to reach a pH of 5.0. The water with the higher pH did not require as much as acid as the one with the lower starting pH. At first glance, this may not make sense. However, note the alkalinity in the center column. Regardless of the starting pH, the higher the alkalinity of the water source, the more acid is required to reduce pH to 5.0. For all water sources, it is the alkalinity that actually determines how much acid to use, not the pH.

How does water alkalinity influence the pH of the growing medium?
In the graph below, vinca plugs were grown for 49 days and constant fed with the same fertilizer at 125 ppm. The plugs were irrigated with three different water sources with varying alkalinities. The pH rose in all three media samples partially due to the fertilizer used and the limestone in the growing medium. However, the higher the alkalinity of the water source, the higher the pH of the growing medium.
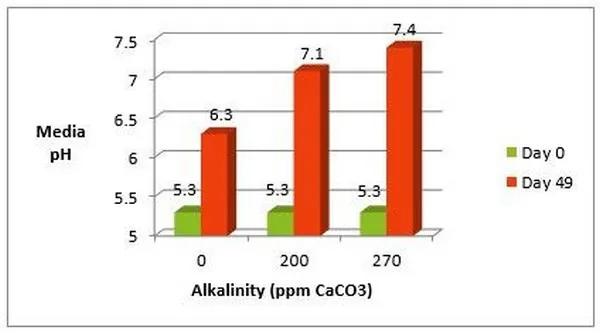
It is clear that the pH of the water and the alkalinity are not the same. In fact, the pH of the water does not dictate the pH of the growing medium, but in fact it is the alkalinity of the water source that influences the pH of the growing medium. This is important for growers to know, since alkalinity has significant impact on growing medium pH, when choosing the correct fertilizer(s) and/or injecting acid.
For more information: Premier Tech
Premier Tech
www.premiertech.com